47 Pages
Preview
Page 1
Aesculap®
2
3
4
5
1
6 7
Aesculap Neurosurgery
USA
de fr es it pt nl da sv fi lv lt ru cs pl sk hu sl hr ro bg tr el
Instructions for use/Technical description Tubular shaft instruments Note for U.S. users This Instructions for Use is NOT intended for United States users. Please discard. The Instructions for Use for United States users can be obtained by visiting our website at www.aesculapusa.com. If you wish to obtain a paper copy of the Instructions for Use, you may request one by contacting your local Aesculap representative or Aesculap's customer service at 1-800-282-9000. A paper copy will be provided to you upon request at no additional cost. Gebrauchsanweisung/Technische Beschreibung Rohrschaftinstrumente Mode d’emploi/Description technique Instruments à chemise Instrucciones de manejo/Descripción técnica Instrumentos de vástago tubular Istruzioni per l’uso/Descrizione tecnica Strumenti a stelo tubolare Instruções de utilização/Descrição técnica Instrumentos tubulares Gebruiksaanwijzing/Technische beschrijving Buisschachtinstrumenten Brugsanvisning/Teknisk beskrivelse Instrumenter med rørskaft Bruksanvisning/Teknisk beskrivning Rörskaftinstrument Käyttöohje/Tekninen kuvaus Putkivartiset instrumentit Lietošanas instrukcijas/tehniskais apraksts Cauruļkāta instrumenti Naudojimo instrukcija/techninis aprašas Vamzdeliniai instrumentai Инструкция по примению/Техническое описание Тубусные инструменты Návod k použití/Technický popis Nástroje s trubkovým dříkem Instrukcja użytkowania/Opis techniczny Instrumenty z trzpieniem rurkowym Návod na použitie/Technický opis Nástroje s rúrovým hriadeľom Használati útmutató/Műszaki leírás Csőszárú műszerek Navodila za uporabo/Tehnični opis Inštrumenti gredi cevi Upute za uporabu/Tehnički opis Cjevasti instrumenti Manual de utilizare/Descriere tehnică Instrumente cu tijă tubulară Упътване за употреба/Техническо описание Инструменти с тръбно тяло Kullanım Kılavuzu/Teknik açiklama Tübüler şaft aletleri Οδηγίες χρήσης/Τεχνική περιγραφή Όργανα με σωληνωτό στέλεχος
9
2
85
10
6 7
1
1
3/10
4/9
6
C
7
2 B
A
1 10
9
8
2 10
8
9
3 2
Aesculap AG | Am Aesculap-Platz | 78532 Tuttlingen | Germany Phone +49 (0) 7461 95-0 | Fax +49 (0) 7461 95-26 00 | www.aesculap.com
3/10
4/9
1
Aesculap® – a B. Braun brand TA013253
2020-06
V6
Change No. 60792
4 4/9
45°
en
5
8
2
1
6
D
7 A
5
en ®
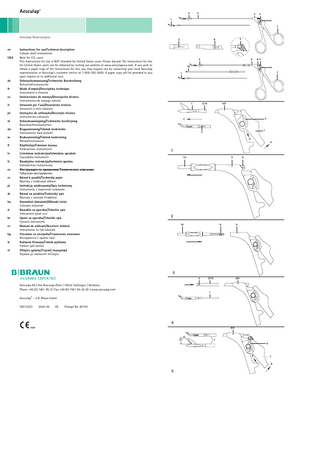
Aesculap Tubular shaft instruments Legend 1 Working insert 2 Lock (working insert/tube) 3 Tube (fixed) or (non-detachable) 4 Fixed handle (tubular shaft instrument) 5 Connection (working insert/movable handle part) 6 Catch (handle/movable handle part) 7 Movable handle 8 Lock (tube/handle) 9 Fixed handle (tubular shaft instrument) 10 Tube
Symbols on product and packages Caution, general warning symbol Caution, see documentation supplied with the product
1.
Intended use
The tubular shaft instruments are used in microneurosurgery, especially for operations in narrow spaces. They are used for cutting, grasping, dissecting etc. of blood vessels and tissue.
2.
Indications
Indications, see Intended use. Note The manufacturer is not responsible for any use of the product against the specified indications and/or the described applications.
3.
Contraindications
None known.
4.
Risks and side effects
As part of the legal obligation to provide information, the possible risks and side effects in connection with the use of surgical instruments known to the manufacturer are highlighted below. These are predominantly process-specific, not product-specific, and include unwanted damage to surrounding tissue, such as bleeding, infection, incompatibility of materials, or parts of instruments left unnoticed in the patient.
5.
Safe handling and preparation
► Ensure that the product and its accessories are operated and used only by persons with the requisite training,
knowledge, or experience. ► Read, follow, and keep the instructions for use. ► Use the product only in accordance with its intended use, see Intended use. ► Remove the transport packaging and clean the new product, either manually or mechanically, prior to its initial
sterilization. ► Store any new or unused products in a dry, clean, and safe place. ► Prior to each use, inspect the product for loose, bent, broken, cracked, worn, or fractured components. ► Do not use the product if it is damaged or defective. Set aside the product if it is damaged. ► Replace any damaged components immediately with original spare parts.
6.
Safe operation
WARNING Risk of injury and/or malfunction! ► Always carry out a function check prior to using the product. WARNING Risk of injury when using the product beyond the field of view! ► Apply the product only under visual control.
7.
Disassembling
7.1
Separating the handle from the working tip (see Fig. 1)
► Completely unlock moveable handle part 7 in the direction of arrow A. ► Pull down movable handle part 7, against the resistance of catch 6, in the direction of arrow B. ► Pull working insert 1 against the resistance of the lock 2 in the direction of arrow C out of handle 4 or 9.
7.2
Separating the handle from the tube (see Fig. 2)
9.
Validated reprocessing procedure
9.1
General safety notes
Note Adhere to national statutory regulations, national and international standards and directives, and local, clinical hygiene instructions for reprocessing. Note For patients with Creutzfeldt-Jakob disease (CJD), suspected CJD, or possible variants of CJD, observe the relevant national regulations concerning the reprocessing of products. Note It should be noted that successful reprocessing of this medical device can only be guaranteed following prior validation of the reprocessing method. The operator/reprocessing technician is responsible for this. The specified chemistry was used for validation.
9.2
General information
Dried and hardened or clotted surgical residues can complicate cleaning or render it ineffective and cause corrosion. Therefore, do not exceed a period of 1h between use and processing, make sure pre-cleaning temperatures are <45°C to prevent clotting, and do not use disinfectants that can cause clotting (active ingredient: aldehyde, alcohol). Excessive neutralizing agents or basic cleaners may result in a chemical attack and/or fading and the laser marking becoming unreadable either visually or by machine. Residues containing chlorine or chlorides, e.g., in surgical residues, medicines, saline solutions, and in the service water used for cleaning, disinfection, and sterilization, will cause corrosion damage (pitting, stress corrosion) and result damage to metallic products. These must be removed by rinsing thoroughly with demineralized water and then drying. Perform additional drying, if necessary. Only process chemicals that have been tested and approved (e.g. VAH or FDA approval or CE mark) and which are compatible with the product’s materials according to the chemical manufacturers’ recommendations may be used for processing the product. All the chemical manufacturer's application specifications must be strictly observed. Failure to do so can result in the following problems: ■ Material damage such as corrosion, cracks, fracturing, premature aging, or swelling. ► Do not use metal cleaning brushes or other abrasives that would damage the product surface and could cause corrosion ► For detailed information on hygienically safe and material-friendly/careful processing, please refer to www.a-k-i.org Header "AKI Brochures", "Red Brochure".
9.3
Reusable products
There are no known effects of processing resulting in damage to the product. The product can be re-used up to 75 times with proper care and if it is undamaged and clean. Any additional reuse falls under the responsibility of the user. Careful visual and functional testing prior to next use is the best way to identify a malfunctioning product.
9.4
Preparations at the place of use
► If applicable, rinse surfaces that are not visible, preferably using deionized water – for instance from disposable
syringes. ► Remove any visible surgical residues as comprehensively as possible using a damp, lint-free cloth. ► Place the wet product in a sealed waste container and take it to be cleaned and disinfected within 1h.
9.5
Preparation before cleaning
► Dismantle the product prior to cleaning, see Disassembling.
9.6
Cleaning/disinfection
9.6.1
Product-specific safety notes on the reprocessing procedure
DANGER Danger to the patient! ► Reprocess the product only with manual pre-cleaning followed by mechanical cleaning. DANGER Risk to patient due to cross contamination! ► Do not clean contaminated products together with uncontaminated products in a tray. CAUTION Damage to the product due to inappropriate cleaning/disinfecting agents and/or excessive temperatures! ► Use cleaning and disinfecting agents approved for aluminum, plastics, and high-grade steel for instance, according to the manufacturer’s instructions. ► Observe specifications regarding concentration, temperature and exposure time. ► Do not exceed the maximum permitted cleaning temperature of 60 °C.
9.7
Mechanical cleaning/disinfection with manual pre-cleaning
Note As a matter of principle, the efficacy of the cleaning and disinfection device must be certified (e.g. in accordance with EN ISO 15883). Note The cleaning and disinfection machine used for processing must be serviced and checked at regular intervals.
9.7.1
Manual pre-cleaning with a brush
Phase
Step
T [°C/°F]
t [min]
Conc. [%]
Water quality
Chemicals
I
Cleaning
RT (cold)
>15
1
D–W
B. Braun Stabimed® fresh
II
Rinsing
RT (cold)
1
-
D–W
-
► Extract tube 10 against the resistance of lock 8 from handle 9.
8.
Assembling
8.1
Connecting the handle and the tube (see Fig. 3)
► Insert tube 10 against the resistance of lock 8 into handle 9. ► Push tube 10 into handle 9 down to the positive stop and engage lock 8.
8.2
Connecting the working insert to the tube (see Fig. 4)
► Insert working insert 1 in tube 3 or 10. ► Push working insert 1 down to lock 2 and engage it against the resistance of lock 2.
8.3
Connecting the handle and the working insert (see Fig. 5)
► Completely unlock moveable handle part 7 in the direction of arrow A. ► Completely open the jaws of working insert 1 (approx. 45°). ► Push movable handle part 7 upwards against the resistance of catch 6, in the direction of arrow D. ► Carry out a function test.
D–W: RT:
Drinking water Room temperature
Phase I ► Use an ultrasonic cleaner (frequency 35kHz) to clean the product for at least 15 min. Make sure all accessible surfaces are wetted and acoustic shadows are prevented. ► Clean the product with a suitable cleaning brush in the solution until all discernible residues have been removed. ► If applicable, brush non-visible surfaces for at least 1 minute with a suitable cleaning brush. ► Mobilize non-rigid components, such as set screws and hinges, during cleaning. ► Then flush these areas thoroughly at least five times with the cleaning disinfectant solution using a disposable syringe (20 ml). Phase II ► Rinse/flush the instrument thoroughly (all accessible surfaces) under running water. ► Mobilize non-rigid components, such as set screws and hinges, during rinsing.
9.7.2
Mechanical alkaline cleaning and thermal disinfection
Type of device: Single-chamber cleaning/disinfection device without ultrasound1) Phase
Step
T [°C/°F]
t [min]
Water quality
Chemicals
I
Pre-rinse
<25/77
3
D–W
-
II
Cleaning
55/131
10
DI–W
Dr. Weigert neodisher® SeptoClean 1 % Working solution2)
III
Neutralization
>10/50
2
DI–W
B. Braun Helimatic® Neutralizer C Working solution 0.15 %
IV
Intermediate rinse I
>10/50
1
DI–W
-
V
Intermediate rinse II
>10/50
1
DI–W
-
VI
Thermal disinfection
90/194
5
DI–W
-
VII
Drying
-
-
-
In accordance with the program for the cleaning and disinfecting machine
D–W: Drinking water DI–W: Deionized water 1) The cleaning and disinfection devices listed below were used to verify cleanability: Miele 7836 CD 2) Prion-deactivating detergent (see Specifications Dr. Weigert neodisher® SeptoClean) ► Place the product in a tray that is suitable for cleaning (avoiding rinsing blind spots) ► Place instruments in the tray with their hinges open. ► Connect components with lumens and channels directly to the rinsing port of the injector carriage. ► Check visible surfaces for residues after mechanical cleaning/disinfecting.
9.8
Inspection, maintenance and checks
CAUTION Damage (metal seizure/friction corrosion) to the product caused by insufficient lubrication! ► Prior to function checks, lubricate moving parts (e.g. joints, pusher components and threaded rods) with maintenance oil suitable for the respective sterilization process (e.g. for steam sterilization: Aesculap STERILIT® I oil spray JG600 or STERILIT® I drip lubricator JG598). ► Allow the product to cool down to room temperature. ► After each complete cleaning, disinfecting and drying cycle, check that the product is dry, clean, operational, and
free of damage (e.g. broken insulation or corroded, loose, bent, broken, cracked, worn, or fractured components). ► Dry the product if it is wet or damp. ► Repeat cleaning and disinfection of products that still show impurities or contamination. ► Check that the product functions correctly. ► Immediately put aside damaged or inoperative products and send them to Aesculap Technical Service, see Tech-
nical Service.
9.9
Packaging
► Appropriately protect products with fine working tips. ► Store products with ratchet locks fully opened or locked no further than in the first notch. ► Place the product in its holder or on a suitable tray. Ensure that all cutting edges are protected. ► Pack trays appropriately for the intended sterilization process (e.g. in Aesculap sterile containers). ► Ensure that the packaging provides sufficient protection against recontamination of the product during storage.
9.10 Steam sterilization Note The product may only be sterilized when dismantled. ► Check to ensure that the sterilizing agent will come into contact with all external and internal surfaces (e.g. by
opening any valves and faucets). ► Validated sterilization process
– Disassemble the product – Steam sterilization using fractional vacuum process – Steam sterilizer according to DIN EN 285 and validated according to DIN EN ISO 17665 – Sterilization using fractional vacuum process at 134 °C, holding time 18 minutes for prion inactivation ► When sterilizing several products at the same time in a steam sterilizer, ensure that the maximum load capacity of the steam sterilizer specified by the manufacturer is not exceeded.
9.11 Storage ► Store sterile products in germ-proof packaging, protected from dust, in a dry, dark, temperature-controlled area.
10.
Technical Service
WARNING Risk of injury and/or malfunction! ► Do not modify the product. ► For service and repairs, please contact your national B. Braun/Aesculap agency.
Modifications carried out on medical technical equipment may result in loss of guarantee/warranty rights and forfeiture of applicable licenses. Service addresses Aesculap Technischer Service Am Aesculap-Platz 78532 Tuttlingen / Germany Phone: +49 (7461) 95-1602 Fax: +49 (7461) 16-5621 E-Mail: ats@aesculap.de Other service addresses can be obtained from the address indicated above.
11.
Disposal
► Adhere to national regulations when disposing of or recycling the product, its components and its packaging!
TA013253
2020-06
V6
Change No. 60792